ISSUE1752
- Jean-Marie Pflomm, Pharm.D., Editor in Chief has disclosed no relevant financial relationships.
- Michael Viscusi, Pharm.D., Associate Editor has disclosed no relevant financial relationships.
- Review the efficacy and safety of etripamil nasal spray (Cardamyst) for treatment of paroxysmal supraventricular tachycardia.
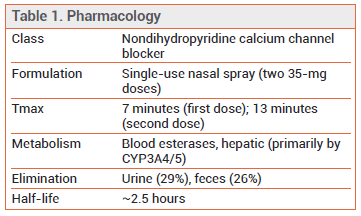
- Description: A nasal spray formulation of a nondihydropyridine calcium channel blocker structurally related to verapamil
- Indication: Conversion of acute symptomatic episodes of paroxysmal supraventricular tachycardia (PSVT) to sinus rhythm in adults
- Efficacy: In a double-blind trial, symptomatic PSVT episodes that had not responded to vagal maneuvers were converted to normal sinus rhythm more often with etripamil than with placebo.
- Adverse Effects: Nasal discomfort and congestion, rhinorrhea, epistaxis, and throat irritation were most common. Clinically significant hypotension has occurred.
- Dosage: One spray in each nostril (70 mg) at symptom onset; can repeat one time 10 minutes later if symptoms persist.
- Cost: One carton containing two single-use devices costs $1649.
- Conclusion: Cardamyst provides a rapid-acting option for outpatient self-treatment of PSVT. The drug is expensive, but it may reduce the need for acute treatment in an emergency department, which would cost more.
The FDA has approved Cardamyst (Milestone), a nasal spray formulation of the nondihydropyridine calcium channel blocker etripamil, for conversion of acute symptomatic episodes of paroxysmal supraventricular tachycardia (PSVT) to sinus rhythm in adults. Cardamyst is the first product containing etripamil to be approved in the US. It is being marketed as the only FDA-approved, self-administered, rapid-acting treatment option for acute episodes of PSVT.

THE DISORDER — PSVT describes any of several regular, generally narrow-QRS complex tachycardias with sudden onset and termination. PSVTs commonly involve reentry within the AV node or between the AV node and an accessory pathway. Symptoms can include palpitations, dyspnea, chest discomfort, and, rarely, syncope. About 1 in 300 persons in the US are thought to have PSVT; older adults and females are at increased risk.1
Vagal maneuvers such as the modified Valsalva maneuver and carotid sinus massage are recommended for acute management of PSVT. If vagal maneuvers fail to terminate the tachycardia, IV adenosine can be used. Hemodynamically stable patients with PSVT that persists after administration of adenosine can receive IV treatment with a nondihydropyridine calcium channel blocker such as diltiazem or verapamil (except patients with systolic heart failure) or a cardioselective beta blocker such as esmolol or metoprolol.2,3 Single oral doses of diltiazem and/or propranolol (both off-label) have been used for outpatient self-treatment of infrequent recurrent episodes of PSVT.4,5 Hemodynamically unstable patients and those with tachycardia that is unresponsive to pharmacologic treatment should undergo electrical cardioversion.2,3
PHARMACOLOGY — Etripamil is structurally related to verapamil. Like verapamil and diltiazem, it inhibits the influx of calcium ions across the membranes of cardiac cells during depolarization, decreasing the velocity of AV nodal conduction and disrupting reentry. The time to peak plasma concentration (Tmax) is much shorter following administration of a single intranasal dose of etripamil (7 minutes) than after an oral dose of a calcium channel blocker (2-4 hours with diltiazem).6
CLINICAL STUDIES — FDA approval of Cardamyst was based on the results of a double-blind trial (RAPID) in 692 adults with a history of PSVT. Patients were randomized to treat symptomatic PSVT episodes that did not respond to vagal maneuvers with up to two self-administered intranasal doses of etripamil or placebo. Among the 184 patients who experienced a confirmed PSVT episode, conversion to sinus rhythm for ≥30 seconds within 30 minutes after the first dose, the primary endpoint, occured more frequently in patients treated with etripamil than with placebo (64% vs 31%; number needed to treat [NNT] 3.0). The median time to conversion was 17.2 minutes with etripamil and 53.5 minutes with placebo.7
ADVERSE EFFECTS — The most common adverse effects of etripamil nasal spray in clinical trials were nasal discomfort, nasal congestion, throat irritation, rhinorrhea, and epistaxis. Syncope could theoretically occur with use of etripamil; clinically significant hypotension was reported in 0.4% of patients in clinical trials. Etripamil has not been shown to prolong the QT interval at clinically relevant doses.
CONTRAINDICATIONS — Etripamil is contraindicated for use in patients with NYHA class II-IV heart failure, Mobitz type II second- or third-degree atrioventricular block, sick sinus syndrome (except in patients with a functioning pacemaker), Wolff-Parkinson-White or Lown-Ganong-Levine syndrome, or manifest pre-excitation (delta wave) on a 12-lead ECG.
PREGNANCY AND LACTATION — Etripamil has not been studied in pregnant or lactating women. In pregnant rats and rabbits, exposure to the drug was not associated with fetal toxicity, except at very high doses that also caused maternal toxicity. Verapamil is secreted into human breast milk in low levels and is generally considered safe for use by women who are pregnant or breastfeeding.8,9 The manufacturer recommends that lactating women pump and discard their milk for 12 hours after use of etripamil.
DOSAGE, ADMINISTRATION, AND COST — Cardamyst is supplied in single-use nasal spray devices that deliver two sprays, each containing 35 mg of etripamil. The recommended dosage is 70 mg (one spray in each nostril) administered at symptom onset. If symptoms persist, a second 70-mg dose can be administered 10 minutes later using a second device. Patients should not use more than two 70-mg doses in a 24-hour period. One carton containing two nasal spray devices costs $1649.10
CONCLUSION — Self-treatment with Cardamyst, a nasal spray formulation of the calcium channel blocker etripamil, was effective in terminating acute episodes of paroxysmal supraventricular tachycardia (PSVT) in approximately two-thirds of treated patients in a placebo-controlled trial. It is likely more rapidacting than oral diltiazem or metoprolol for such use, but comparative trials are not available. The drug is expensive, but it may reduce the need for acute treatment in an emergency department, which would cost more.
- M Rehorn et al. Prevalence and incidence of patients with paroxysmal supraventricular tachycardia in the United States. J Cardiovasc Electrophysiol 2021; 32:2199. doi:10.1111/jce.15109
- RL Page et al. 2015 ACC/AHA/HRS guideline for the management of adult patients with supraventricular tachycardia: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. J Am Coll Cardiol 2016; 67:e27. doi:10.1016/j.jacc.2015.08.856
- J Brugada et al. 2019 ESC guidelines for the management of paients with supraventricular tachycardia. Eur Heart J 2020; 41:655. doi:10.1093/eurheartj/ehz467
- P Alboni et al. Efficacy and safety of out-of-hospital self-administered single-dose oral drug treatment in the management of infrequent, well-tolerated paroxysmal supraventricular tachycardia. J Am Coll Cardiol 2001; 37:548. doi:10.1016/s0735-1097(00)01128-1
- SJ Yeh et al. Termination of paroxysmal supraventricular tachycardia with a single oral dose of diltiazem and propranolol. Circulation 1985; 71:104. doi:10.1161/01.cir.71.1.104
- JE Ip et al. Pharmacokinetics and pharmacodynamics of etripamil, an intranasally administered, fast-acting, nondihydropyridine calcium channel blocker. Clin Pharmacol Drug Dev 2024; 13:367. doi:10.1002/cpdd.1383
- BS Stambler et al. Self-administered intranasal etripamil using a symptom-prompted, repeat-dose regimen for atrioventricular-nodal-dependent supraventricular tachycardia (RAPID): a multicentre, randomised trial. Lancet 2023; 402:118. doi:10.1016/s0140-6736(23)00776-6
- GG Briggs et al. Briggs drugs in pregnancy and lactation: a reference guide to fetal and neonatal risk. 12th edition. Philadelphia: Lippincott Williams & Wilkins, 2021.
- Drugs and Lactation Database (LactMed) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-. Verapamil. [Updated 2025 Aug 15]. Available at: https://bit.ly/4bu0icl. Accessed March 26, 2026.
- Approximate WAC. WAC = wholesaler acquisition cost or manufacturer's published price to wholesalers; WAC represents a published catalogue or list price and may not represent an actual transactional price. Source: AnalySource® Monthly. March 5, 2026. Reprinted with permission by First Databank, Inc. All rights reserved. ©2026. www.fdbhealth.com/drug-pricing-policy.
The Medical Letter, Inc. does not warrant that all the material in this publication is accurate and complete in every respect. The Medical Letter, Inc. and its editors shall not be held responsible for any damage resulting from any error, inaccuracy, or omission.