ISSUE1752
- Jean-Marie Pflomm, Pharm.D., Editor in Chief has disclosed no relevant financial relationships.
- Susan Daron, Pharm D., Associate Editor has disclosed no relevant financial relationships.
- Review the efficacy and safety of pivmecillinam (Pivya) for treatment of uncomplicated urinary tract infections in women.
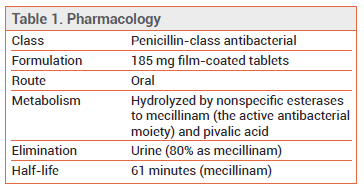
- Description: An oral prodrug of the penicillin antibiotic mecillinam
- Indication: Treatment of uncomplicated urinary tract infection (uUTI) caused by susceptible isolates of Escherichia coli, Proteus mirabilis, or Staphylococcus saprophyticus in adult females
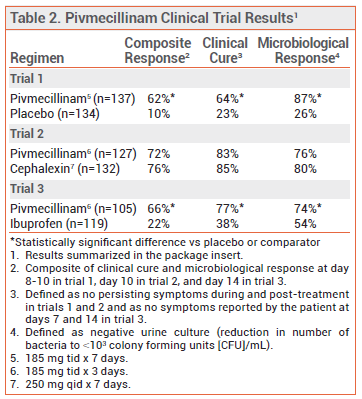
- Efficacy: In a clinical trial in adult women, pivmecillinam was similar in efficacy to cephalexin for treatment of uUTI.
- Adverse Effects: Most common are nausea, diarrhea, vulvovaginal candidiasis, genital pruritus, and headache. Contraindicated in patients with porphyria.
- Drug Interactions: Use with valproic acid or other pivalate-generating drugs should be avoided. May reduce clearance of methotrexate.
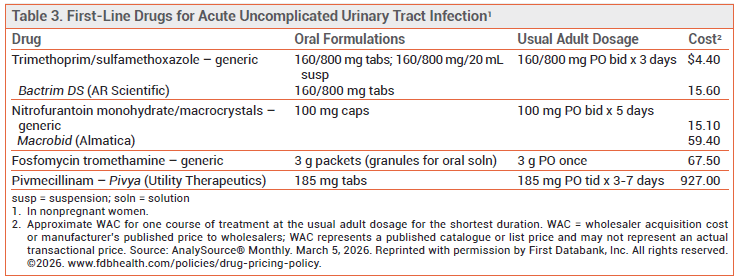
- Dosage: 185 mg PO tid x 3-7 days
- Cost: A 3-day course of treatment costs $927.
- Conclusion: Pivmecillinam is a recommended first-line option for treatment of acute uUTI in adult women, but it is dosed more frequently and is more expensive than other first-line drugs.
The FDA has approved pivmecillinam (Pivya – Utility Therapeutics), an oral penicillin-class antibacterial drug, for treatment of uncomplicated urinary tract infections (uUTIs) caused by susceptible isolates of Escherichia coli, Proteus mirabilis, or Staphylococcus saprophyticus in adult females. Pivmecillinam was approved by the FDA in 2024 but only recently became available in the US. It has been used in Europe for over 40 years.

ACUTE uUTI — The most common bacterial cause of acute uUTI in otherwise healthy nonpregnant women is E. coli; other common pathogens include Klebsiella pneumoniae, P. mirabilis, and S. saprophyticus. Infections caused by drug-resistant E. coli, including extended-spectrum beta-lactamase (ESBL)-producing strains, have been increasing worldwide.1
STANDARD TREATMENT ― Recommended first-line options for empiric treatment of uUTI in nonpregnant women include trimethoprim/sulfamethoxazole, nitrofurantoin, fosfomycin, and pivmecillinam (see Table 3).2 Amoxicillin/clavulanate, cefpodoxime, and cefadroxil are second-line options. The fluoroquinolones ciprofloxacin and levofloxacin are alternatives for patients who cannot take a first- or second-line antibiotic. Increasing resistance of common uropathogens to all of these drugs is a concern.
The oral triazaacenaphthylene antibiotic gepotidacin (Blujepa) and an oral fixed-dose combination of the thiopenem antibacterial drug sulopenem etzadroxil and the renal tubule transport inhibitor probenecid (Orlynvah) were recently approved by the FDA for treatment of uUTI.3,4 They are not recommended for empiric treatment of acute uUTI.
MECHANISM OF ACTION ― Pivmecillinam is a prodrug of mecillinam, a beta-lactam antibiotic that interferes with bacterial cell wall biosynthesis. Unlike most other beta-lactams, which bind to gram-negative penicillin-binding protein (PBP)-1 and PBP-3, pivmecillinam preferentially binds to PBP-2.
SPECTRUM OF ACTIVITY ― Pivmecillinam has demonstrated activity in clinical uUTI against E. coli, P. mirabilis, and S. saprophyticus. Mecillinam has demonstrated in vitro activity against some ESBL-producing Enterobacterales. Resistance of E. coli to pivmecillinam has remained low in countries where the drug is used routinely for treatment of UTI.5,6
CLINICAL STUDIES ― FDA approval of pivmecillinam was based on the results of three randomized, double-blind trials that compared the drug to placebo, cephalexin, or ibuprofen in adult females with uUTI.7-9 In all three trials, the most common baseline pathogen was E. coli. Rates of composite response (clinical cure and microbiological response) with pivmecillinam were significantly higher than those with placebo or ibuprofen and were similar to those with cephalexin (see Table 2).
In another randomized, double-blind trial in women with uUTI, rates of bacterial cure (75% vs 91%) and clinical cure (82% vs 88%) were lower with pivmecillinam than with norfloxacin.10
ADVERSE EFFECTS ― The most common adverse effects of pivmecillinam in the placebo-controlled trial were nausea, diarrhea, vulvovaginal candidiasis, genital pruritus, and headache.
Serious hypersensitivity reactions and severe cutaneous adverse reactions, including acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), Stevens-Johnson syndrome (SJS), and toxic epidermal necrolysis (TEN), have been reported with pivmecillinam. Cross-hypersensitivity has been reported with other beta-lactams, including penicillins, cephalosporins, and carbapenems.
Carnitine depletion may occur with pivalate-containing compounds such as pivmecillinam; the drug is contraindicated for use in patients with disorders known to cause carnitine depletion. Clinical effects of decreased carnitine have not been reported with short courses of pivmecillinam. Alternative antibacterial drugs should be considered in patients at risk for reductions in serum carnitine, such as those with significant renal impairment or decreased muscle mass.
Pivmecillinam has been associated with acute attacks of porphyria and is contraindicated for use in patients with porphyria. As with other antibiotics, use of pivmecillinam could result in Clostridioides difficile-associated diarrhea.
DRUG INTERACTIONS — Concurrent use of pivmecillinam and valproic acid or other pivalate-generating drugs increases the risk of carnitine depletion and should be avoided. Penicillin-class antibiotics can reduce the clearance of methotrexate.
PREGNANCY AND LACTATION ― Data on the use of pivmecillinam in pregnant women are limited. In observational studies, first-trimester use of the drug was not associated with an increased risk of major birth defects. In animal studies, there was no evidence of embryofetal toxicity at doses up to 5-fold higher than the maximum recommended daily human dose. False-positive tests for isovaleric acidemia, a rare genetic condition that inhibits proper breakdown of leucine-containing proteins, are possible in infants born to mothers treated with pivmecillinam prior to delivery.
Limited data suggest that low levels of pivmecillinam are present in human milk.11 Rash and diarrhea have been reported in breastfed infants exposed to mecillinam.
DOSAGE AND ADMINISTRATION ― The recommended dosage of Pivya for uUTI is 185 mg orally three times daily for 3 to 7 days.
CONCLUSION ― Pivmecillinam (Pivya), an oral penicillin-class antibiotic that is recommended as a first-line option for empiric treatment of uUTI in nonpregnant women, is now available in the US. It is dosed more frequently and costs much more than other first-line options. Resistance of Escherichia coli to pivmecillinam appears to be uncommon.
- PD Tamma et al. Infectious Diseases Society of America 2024 guidance on the treatment of antimicrobial-resistant gramnegative infections. Clin Infect Dis 2024 August 7 (epub). doi:10.1093/cid/ciae403
- K Gupta et al. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: a 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis 2011; 52:e103. doi:10.1093/cid/ciq257
- Gepotidacin (Blujepa) – a new antibiotic for uncomplicated UTI. Med Lett Drugs Ther 2026; 68:1.
- Sulopenem etzadroxil/probenecid (Orlynvah) for uncomplicated UTIs. Med Lett Drugs Ther 2025; 67:179.
- N Frimodt-Møller et al. Pivmecillinam, the paradigm of an antibiotic with low resistance rates in Escherichia coli urine isolates despite high consumption. J Antimicrob Chemother 2022; 78:289. doi:10.1093/jac/dkac396
- TP Lodise et al. Review of the in vitro microbiological activity of mecillinam against common uropathogens in uncomplicated urinary tract infection: focus on resistant pathogens. Open Forum Infect Dis 2024; 11:ofae296. doi:10.1093/ofid/ofae296
- SA Ferry et al. Clinical and bacteriological outcome of different doses and duration of pivmecillinam compared with placebo therapy of uncomplicated lower urinary tract infection in women: The LUTIW project. Scand J Prim Health Care 2007; 25:49. doi:10.1080/02813430601183074
- AP Menday. Comparison of pivmecillinam and cephalexin in acute uncomplicated urinary tract infection. Int J Antimicrob Agents 2000; 13:183. doi:10.1016/s0924-8579(99)00118-1
- I Vik et al. Ibuprofen versus pivmecillinam for uncomplicated urinary tract infection in women – a double-blind, randomized non-inferiority trial. PLoS Med 2018; 15:e1002569. doi:10.1371/journal.pmed.1002569
- LE Nicolle et al. Three days of pivmecillinam or norfloxacin for treatment of acute uncomplicated urinary infection in women. Scand J Infect Dis 2002; 34:487. doi:10.1080/00365540110080728
- Drugs and Lactation Database (LactMed) [Internet]. Bethesda (MD): National Institute of Child Health and Human Development; 2006-. Amdinocillin. [Updated 2023 May 15].Available at: https://bit.ly/4hOdkm4. Accessed March 26, 2026.
The Medical Letter, Inc. does not warrant that all the material in this publication is accurate and complete in every respect. The Medical Letter, Inc. and its editors shall not be held responsible for any damage resulting from any error, inaccuracy, or omission.