ISSUE1753
- Jean-Marie Pflomm, Pharm.D., Editor in Chief has disclosed no relevant financial relationships.
- Michael Viscusi, Pharm.D., Associate Editor has disclosed no relevant financial relationships.
- Explain the current approach to the management of patients with sexually transmitted infections.
- Discuss the pharmacologic agents available for treatment of sexually transmitted infections and compare them based on their efficacy, dosage and administration, and potential adverse effects.
- Determine the most appropriate therapy given the clinical presentation of an individual patient with a sexually transmitted infection.
- Partner Treatment
- Post-Exposure Prophylaxis
- Chlamydia
- Nongonococcal Urethritis and Cervicitis
- Gonorrhea
- Epididymitis
- Proctitis
- Trichomoniasis
- Bacterial Vaginosis
- Pelvic Inflammatory Disease
- Syphilis
- Genital Herpes
- Anogenital Warts
- Pediculosis Pubis and Scabies
- Drug Adverse Effects
- Drug Interactions
- References
This review includes recommendations for the treatment of common sexually transmitted infections (STIs) other than HIV and viral hepatitis in nonpregnant adolescents and adults. Some of the indications and dosages included here have not been approved by the FDA.
PARTNER TREATMENT — Sex partners of patients with an STI should be examined and tested for STIs if possible. Alternatively, clinicians can provide treatment for sex partners of their patients without direct examination or testing by either writing a prescription for the partner or giving the partner's medication to their patient, a practice called expedited partner therapy (EPT).1 EPT is legally permissible or potentially allowable in all 50 US states.2
POST-EXPOSURE PROPHYLAXIS — In randomized, open-label trials in men who have sex with men (MSM) and transgender women who had a bacterial STI within the previous 12 months, post-exposure prophylaxis (PEP) with a single 200-mg dose of doxycycline taken within 72 hours after condomless sex decreased the incidence of chlamydia, syphilis, and (in some studies) gonorrhea. Data on the efficacy of doxycycline PEP in females are limited.3
The CDC recommends that MSM and transgender women who had a bacterial STI (chlamydia, syphilis, gonorrhea) within the previous 12 months be offered doxycycline PEP. Doxycycline PEP can be offered to other high-risk patients based on clinical judgment. A single dose of doxycycline 200 mg should be taken as soon as possible (within 72 hours) after condomless oral, anal, or vaginal sex (maximum one dose/day).3,4
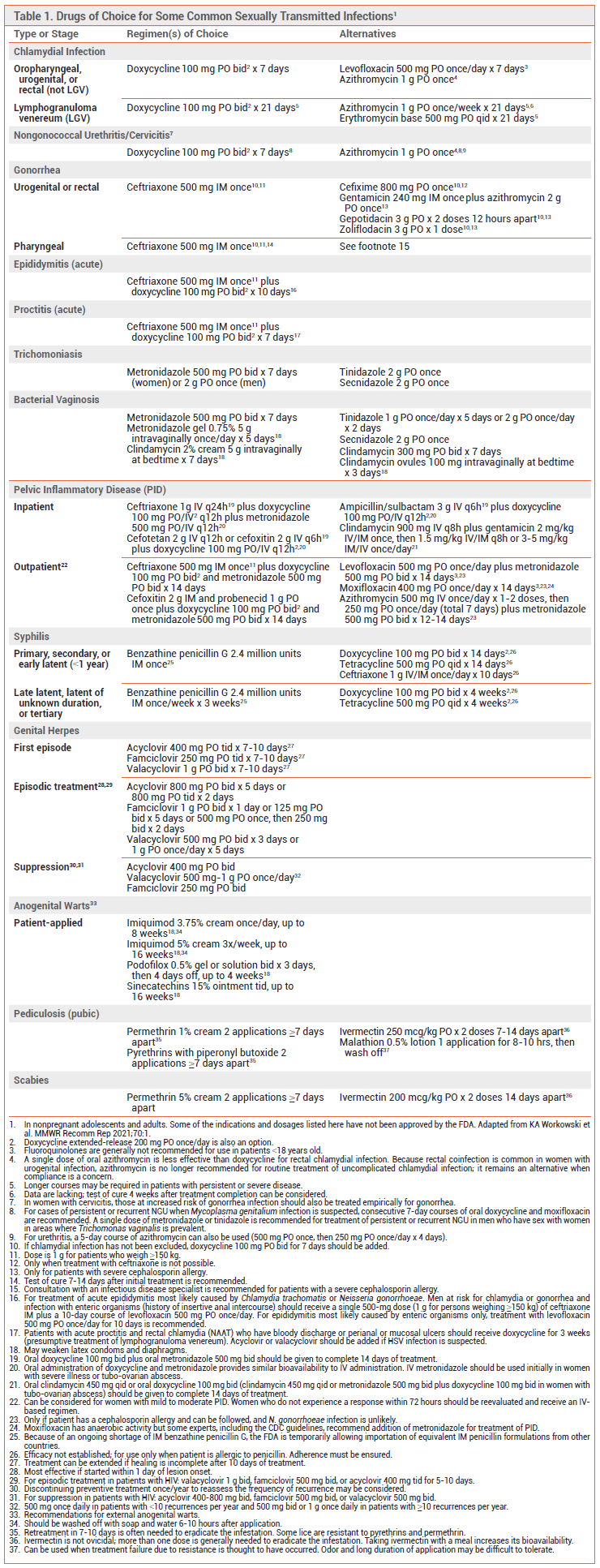
CHLAMYDIA — Chlamydia is the most frequently reported bacterial STI in the US. Asymptomatic infection is common. The treatment of choice for oropharyngeal, urogenital, and rectal Chlamydia trachomatis infection is 7 days of oral doxycycline. A 7-day course of oral levofloxacin is an alternative. A single dose of azithromycin is less effective than doxycycline for treatment of rectal C. trachomatis infection, which is common in women with urogenital infection, but it can be considered if compliance is a concern.5
Lymphogranuloma Venereum – In the US, infections with C. trachomatis serovars that cause lymphogranuloma venereum (LGV; serovars L1-L3) present primarily as proctocolitis, typically in patients with rectal exposure. Patients who present with symptoms consistent with LGV (proctocolitis or genital ulcer with lymphadenopathy) should be offered presumptive treatment.5,6 A 3-week course of oral doxycycline is generally recommended for treatment of LGV, but longer courses may be required in patients with severe or persistent disease.7 A shorter course of oral doxycycline (7-14 days) may be effective in some patients, but data are lacking.8 Oral azithromycin and erythromycin are alternatives.7
Follow-Up – Test of cure is generally not needed in patients who are treated for chlamydia with a recommended or alternative regimen. All patients should be tested for reinfection 3 months after completing treatment.5
Partner Treatment – Sex partners of patients with chlamydia should be offered treatment. EPT is an alternative to in-person screening, but MSM may not be optimal candidates for EPT because they are at increased risk for coexisting infections, especially HIV and syphilis, and data on the effectiveness of EPT in reducing persistent or recurrent infection in this population are limited.1 Asymptomatic partners of persons with LGV should receive presumptive treatment with a 7-day course of doxycycline.5
NONGONOCOCCAL URETHRITIS AND CERVICITIS — No pathogen can be identified in 40-50% of nongonococcal urethritis (NGU) cases in the US. Up to 40% of cases are caused by C. trachomatis and 15-25% are caused by Mycoplasma genitalium. Other pathogens include Trichomonas vaginalis, herpes simplex virus (HSV), and adenovirus. Enteric organisms can cause NGU following insertive anal intercourse.5
Presumptive treatment should be started at the time of diagnosis. The treatment of choice for NGU, including chlamydial urethral and rectal infections, is a 7-day course of oral doxycycline. Oral azithromycin (either a single dose or a 5-day course) is less effective than doxycycline for treatment of rectal C. trachomatis infection. Levofloxacin has limited activity against M. genitalium and is not recommended. Persistent or recurrent NGU in a patient who has not been re-exposed to an untreated sex partner is most commonly caused by M. genitalium; if infection with M. genitalium is suspected, consecutive 7-day courses of oral doxycycline and oral moxifloxacin should be used.5,9 In areas where T. vaginalis is prevalent, a single dose of oral metronidazole or tinidazole is recommended for treatment of persistent or recurrent NGU in men who have sex with women.5
A 7-day course of oral doxycycline is recommended for presumptive treatment of nongonococcal cervicitis; a single dose of oral azithromycin is an alternative. Women at increased risk of gonorrhea infection, such as those who are <25 years old, who live in communities with high rates of gonococcal infection, or who have a new sex partner, a sex partner with concurrent partners, or a sex partner who has an STI, should also receive empiric treatment for gonorrhea.5
GONORRHEA — The treatment of choice for uncomplicated urogenital, rectal, or pharyngeal gonorrhea is a single IM injection of ceftriaxone. Co-infection with C. trachomatis is common; if chlamydial infection has not been excluded, a 7-day course of oral doxycycline should be added.5
When treatment with ceftriaxone is not possible, a single 800-mg dose of oral cefixime can be used to treat uncomplicated urogenital and rectal gonorrhea; the efficacy of the drug for treatment of pharyngeal gonorrhea is limited. In patients who have a severe cephalosporin allergy, a single dose of IM gentamicin plus oral azithromycin can be used, but data on its efficacy for treatment of pharyngeal infection are limited.5
Resistance – The WHO has warned of an increasing global prevalence of gonorrhea strains that are resistant to ceftriaxone and cefixime. Treatment failures have been reported in other countries.10 Cephalosporin resistance resulting in treatment failure remains rare in the US.5,11
New Antibiotics – In 2025, the FDA approved two oral bacterial type II topoisomerase inhibitors for treatment of uncomplicated urogenital gonorrhea: gepotidacin (Blujepa), a triazaacenaphthylene, and zoliflodacin (Nuzolvence), a spiropyrimidenetrione. In randomized trials, microbiological cure rates with two doses of gepotidacin or a single dose of zoliflodacin suspension were noninferior to those with ceftriaxone plus azithromycin (addition of azithromycin is no longer recommended).12,13
Follow-Up – Test of cure is generally not needed for patients who are treated for uncomplicated urogenital or rectal gonorrhea with a recommended or alternative regimen. It is recommended 7-14 days after treatment in patients with pharyngeal gonorrhea, but testing at 7 days may increase the risk of a false positive result. Rescreening 3 months after treatment completion is recommended for all patients with urogenital, rectal, or pharyngeal gonorrhea.5
Partner Treatment – Sex partners of patients with gonorrhea should undergo testing and presumptive treatment for gonorrhea, ideally with IM ceftriaxone. If the partner’s access to prompt clinical evaluation and treatment is limited, EPT with a single 800-mg dose of oral cefixime can be considered. If chlamydial co-infection has not been excluded, 7-day course of doxycycline should be added; a single 1-g dose of azithromycin can be substituted for doxycycline if adherence is a concern, but it is less effective for treatment of rectal chlamydia. MSM may not be optimal candidates for EPT because they are at increased risk for coexisting infections, especially HIV and syphilis, and data on the effectiveness of EPT in reducing persistent or recurrent infection in this population are limited.5,14
EPIDIDYMITIS — For acute epididymitis in sexually active men, which is most frequently caused by C. trachomatis or Neisseria gonorrhoeae, empiric treatment with a single dose of IM ceftriaxone plus oral doxycycline for 10 days is recommended. Enteric organisms may also be present in men who practice insertive anal intercourse; a single dose of IM ceftriaxone plus oral levofloxacin for 10 days is recommended for such patients. In men with epididymitis most likely caused by enteric organisms (e.g., those who have undergone urinary tract instrumentation procedures), levofloxacin monotherapy can be considered.5
PROCTITIS — Acute proctitis can occur following oral, digital, or genital contact. It is commonly caused by N. gonorrhoeae, C. trachomatis (including LGV-causing serovars), Treponema pallidum, or HSV. Syphilitic proctitis has been reported. Empiric treatment with a single dose of IM ceftriaxone plus a 7-day course of oral doxycycline is recommended; if LGV is suspected, doxycycline should be taken for 3 weeks. Acyclovir or valacyclovir should be added if HSV infection is suspected.5 Syphilitic proctitis should be treated with a single IM dose of benzathine penicillin G or, alternatively, with a 14-day course of oral doxycycline.15
TRICHOMONIASIS — A 7-day course of oral metronidazole is recommended for all women with trichomoniasis.5,16 A single oral dose of tinidazole or secnidazole is an alternative; these drugs may be less likely than metronidazole to cause GI adverse effects. Tinidazole and secnidazole may be effective against metronidazole-resistant T. vaginalis.17,18 No studies have conclusively documented the efficacy of any treatment regimen in men, but single-dose oral metronidazole is recommended.5
Follow-Up – Reinfection is common; retesting within 3 months after completing treatment is recommended for all sexually active women.5
Partner Treatment – Sex partners of patients with trichomoniasis should be treated with metronidazole (a 7-day course for women and a single dose for men).5
BACTERIAL VAGINOSIS — In bacterial vaginosis (BV), normal hydrogen peroxide-producing lactobacilli are replaced by overgrowth of various species of bacteria such as Gardnerella spp., Prevotella spp., Fannyhessea vaginae, and Sneathias spp. BV has been associated with an increased risk of STIs, including HIV. BV is not considered a traditional STI, but in a recently published trial, treatment of male sex partners with oral metronidazole and topical clindamycin (applied to the penile skin) for 7 days reduced the rate of recurrence in their female sex partners, suggesting sexual transmission.19,20
Oral or intravaginal metronidazole or intravaginal clindamycin cream is usually effective for treatment of BV. Oral tinidazole, secnidazole, and clindamycin and intravaginal clindamycin ovules are alternatives. BV recurrence is common; retreatment with the same drug or an alternative is usually effective in the short term. Maintenance suppressive therapy with twice-weekly intravaginal metronidazole gel can reduce recurrence rates in women with frequent symptomatic recurrences.5
PELVIC INFLAMMATORY DISEASE — C. trachomatis and N. gonorrhoeae can cause acute pelvic inflammatory disease (PID). M. genitalium has also been associated with PID pathogenesis. Secondary pathogens include Mycoplasma hominis, Haemophilus spp., and various facultative and anaerobic bacteria. Treatment regimens should include broad-spectrum antimicrobial coverage of likely pathogens, including C. trachomatis and N. gonorrhoeae.
Recommended initial regimens for inpatient treatment include IV ceftriaxone plus doxycycline and metronidazole (for anaerobic coverage), IV cefotetan plus doxycycline, and IV cefoxitin plus doxycycline. Oral doxycycline and metronidazole are recommended to complete 14 days of treatment. Ampicillin/sulbactam plus doxycycline and clindamycin plus gentamicin are alternative parenteral regimens.
Patients with mild to moderate acute PID can receive outpatient treatment with a single IM dose of a cephalosporin (typically ceftriaxone) plus 2 weeks of oral doxycycline and metronidazole. Oral levofloxacin plus metronidazole, moxifloxacin with or without metronidazole, or azithromycin plus metronidazole can be considered in patients with a cephalosporin allergy if infection with N. gonorrhoeae is unlikely and the patient can be followed. Moxifloxacin is effective for treatment of PID caused by M. genitalium.5,21
SYPHILIS — Parenteral penicillin G remains the treatment of choice for all stages of syphilis.5,22 Primary, secondary, or early latent syphilis (duration <1 year) should be treated with a single IM injection of benzathine penicillin G.23 A 14-day course of oral doxycycline or tetracycline and 10-14 days of treatment with IM or IV ceftriaxone are alternatives. Cefixime may also be effective, but data are limited.24 For late latent syphilis (duration >1 year or unknown) or tertiary syphilis (gumma or cardiovascular), treatment with 3 once-weekly doses of IM benzathine penicillin G is recommended. Oral doxycycline or tetracycline for 4 weeks is an alternative for treatment of late latent syphilis when benzathine penicillin G cannot be given.5
Because of ongoing shortages of IM benzathine penicillin G, the FDA is temporarily allowing importation of equivalent IM penicillin formulations from other countries.25-27
GENITAL HERPES — HSV-2 is the most prevalent cause of recurrent genital herpes, but the majority of new cases are now caused by HSV-1.28 In patients with an initial genital HSV infection, acyclovir, famciclovir, or valacyclovir taken orally for 7-10 days (or longer, if healing is incomplete) can shorten the duration of pain, systemic symptoms, and viral shedding. Episodic treatment of recurrent symptomatic lesions with the same drugs can facilitate healing if started immediately after symptom onset. For prevention of recurrent episodes of genital herpes, valacyclovir has been more effective than famciclovir,29 and it can reduce the risk of HSV transmission to sex partners.30
Persons with HIV infection may need higher doses and/or longer courses of treatment. IV acyclovir is recommended for treatment of HSV infection that is severely symptomatic or associated with complications requiring hospitalization. IV foscarnet is used for treatment of antiviral-resistant herpes.5,28
ANOGENITAL WARTS — Human papillomavirus (HPV) types 6 and 11 are the most common causes of external anogenital warts; persistent infections with other HPV types (16, 18, or others) cause the majority of cancers and precancers in the anogenital tract and oropharynx. Trichloroacetic acid or bichloroacetic acid, cryotherapy with liquid nitrogen or a cryoprobe, and surgical removal are the most widely used provider-administered treatments for external anogenital warts. Imiquimod 3.75% and 5% cream, podofilox 0.5% solution or gel, and sinecatechins 15% ointment can be applied at home. The initial response rate is 60-70% for all treatments except surgical removal; 20-30% of responders will have a recurrence, many of which will respond to a different therapy. No treatment has been shown to eradicate HPV or to modify the risk of cervical dysplasia or cancer in patients with HPV infection.5
PEDICULOSIS PUBIS AND SCABIES — Phthirus pubis (pubic lice) and Sarcoptes scabiei (scabies) can be transmitted by intimate exposure. Topical treatment with permethrin or pyrethrins with piperonyl butoxide is preferred for pubic lice; retreatment 7-10 days after the initial application is often required to eradicate the infestation. Malathion lotion can be used when resistance to permethrin or pyrethrins is suspected, but it is malodorous and requires a longer duration of application. Oral ivermectin is effective, but treatment should be repeated in 7-14 days because ivermectin does not prevent eggs from hatching.5,31 Scabies should be treated with two applications of permethrin cream 7 days apart or with two oral doses of ivermectin taken 14 days apart. Lindane (no longer marketed in the US) should generally be avoided because it can cause CNS toxicity.5
Partner Treatment – Sex partners and others who had close personal contact with the infected person within the last month should be treated.
DRUG ADVERSE EFFECTS — Antibiotics used for treatment of STIs can increase the risk of Clostridioides difficile infection.
Tetracyclines such as doxycycline can cause GI disturbances and photosensitivity. Azithromycin and erythromycin can cause GI disturbances, headache, dizziness, vaginitis, hepatotoxicity, and QT-interval prolongation.32 Penicillins and cephalosporins can cause rash, GI disturbances, allergic reactions, hemolytic anemia, neutropenia, cholestatic hepatitis, serum sickness, and seizures.
Fluoroquinolones can cause GI disturbances, tremors, rash, oral and vaginal Candida infections, eosinophilia, neutropenia, leukopenia, increased aminotransferase and serum creatinine levels, insomnia, photosensitivity reactions, and peripheral neuropathy. They have also been associated with hyperglycemia and severe hypoglycemia, especially in older adults and in those with diabetes. CNS effects including seizures, delirium, agitation, nervousness, and disturbances in attention, memory, and orientation have occurred. Other serious adverse effects include tendinitis, tendon rupture, aortic aneurysm, exacerbation of myasthenia gravis, and QT-interval prolongation and torsades de pointes.32
Oral metronidazole frequently causes a metallic taste, GI disturbances, and headache. Neurologic adverse effects including seizures and neuropathy have been reported, particularly at high doses and with repeated or prolonged use. Tinidazole and secnidazole can cause similar adverse effects, but they may be better tolerated than metronidazole.
Gentamicin can cause renal toxicity; the risk is higher in older patients, those with renal impairment, and those receiving other nephrotoxic drugs. Ototoxicity, neuromuscular blockade, and serious hypersensitivity reactions can also occur.
Gepotidacin can cause GI disturbances, cholinergic adverse effects, and QT-interval prolongation.32 Serious hypersensitivity reactions can occur. Zoliflodacin can cause headache and hypersensitivity reactions.
Oral clindamycin can cause GI adverse effects and rash. Allergic reactions can occur.
Acyclovir is generally well tolerated. GI disturbances, headache, and malaise can occur. Oral acyclovir has been rarely associated with myalgia, rash, Stevens-Johnson syndrome, neutropenia and other hematologic toxicities, tremors, lethargy, confusion, hallucinations, seizures, encephalopathy, and coma. CNS adverse effects are more likely to occur in older patients and in those with renal impairment. Adverse effects of valacyclovir are similar to those with acyclovir. Famciclovir is also generally well tolerated. Headache, nausea, and diarrhea can occur. Thrombocytopenia, confusion, hallucinations, and nephrotoxicity have been reported.
DRUG INTERACTIONS — Coadministration of oral products containing polyvalent cations (e.g., calcium, magnesium, iron) can decrease absorption of oral tetracyclines and fluoroquinolones; administration should be separated by several hours.
Concurrent use of azithromycin, erythromycin, fluoroquinolones, or gepotidacin and other QT-interval-prolonging drugs can result in additive effects and increase the risk of torsades de pointes.32 Erythromycin is a moderate inhibitor of CYP3A4 and can increase serum concentrations of drugs that are metabolized by CYP3A4. Gepotidacin is a substrate and an inhibitor of CYP3A4; use with moderate or strong CYP3A4 inhibitors or with sensitive CYP3A4 substrates should be avoided.33
Use of fluoroquinolones with glucose-lowering drugs may increase the risk of hypoglycemia. Concurrent use of fluoroquinolones and nonsteroidal anti-inflammatory drugs (NSAIDs) may lower the seizure threshold.
- CDC. Expedited partner therapy. July 16, 2024. Available at: https://bit.ly/4sMrieF. Accessed April 9, 2026.
- CDC. Legal status of expedited partner therapy (EPT). June 2, 2025. Available at: https://bit.ly/4m5PK8h. Accessed April 9, 2026.
- In brief: Doxycycline for STI post-exposure prophylaxis. Med Lett Drugs Ther 2024; 66:119.
- LH Bachmann et al. CDC clinical guidelines on the use of doxycycline postexposure prophylaxis for bacterial sexually transmitted infection prevention, United States, 2024. MMWR Recomm Rep 2024; 73:1. doi:10.15585/mmwr.rr7302a1
- KA Workowski et al. Sexually transmitted infections treatment guidelines, 2021. MMWR Recomm Rep 2021; 70:1. doi:10.15585/mmwr.rr7004a1
- JC Dombrowski et al. Doxycycline versus azithromycin for the treatment of rectal chlamydia in men who have sex with men: a randomized controlled trial. Clin Infect Dis 2021; 73:824. doi:10.1093/cid/ciab153
- CE Morales-Múnera et al. AEDV expert document on the management of ulcerative venereal infections. Actas Dermosifiliogr 2025; 116:T159. doi:10.1016/j.ad.2024.11.012
- R Simons et al. Observed treatment responses to short-course doxycycline therapy for rectal lymphogranuloma venereum in men who have sex with men. Sex Transm Dis 2018; 45:406. doi:10.1097/olq.0000000000000772
- RN Werner et al. German evidence- and consensus-based guideline on the management of penile urethritis. J Dtsch Dermatol Ges 2025; 23:254. doi:10.1111/ddg.15617
- WHO. Enhanced Gonococcal Antimicrobial Surveillance Programme (EGASP): gonorrhoea treatment optimization, 2024 report. November 19, 2025. Available at: https://bit.ly/3NTBD9e. Accessed April 9, 2026.
- CDC. Drug-resistant gonorrhea. February 15, 2024. Available at: https://bit.ly/4rOEy0Q. Accessed April 9, 2026.
- JDC Ross et al. Oral gepotidacin for the treatment of uncomplicated urogenital gonorrhoea (EAGLE-1): a phase 3 randomised, open-label, non-inferiority, multicentre study. Lancet 2025; 405:1608. doi:10.1016/s0140-6736(25)00628-2
- A Luckey et al. Zoliflodacin versus ceftriaxone plus azithromycin for treatment of uncomplicated urogenital gonorrhoea: an international, randomised, controlled, open-label, phase 3, non-inferiority clinical trial. Lancet 2026; 407:147. doi:10.1016/s0140-6736(25)01953-1
- CDC. Clinical treatment of gonorrhea. July 11, 2024. Available at: https://bit.ly/47jATRr. Accessed April 9, 2026.
- M Struyve et al. Primary syphilitic proctitis: case report and literature review. Acta Gastroenterol Belg 2018; 81:430.
- PJ Kissinger et al. Diagnosis and management of Trichomonas vaginalis: summary of evidence reviewed for the 2021 Centers for Disease Control and Prevention sexually transmitted infections treatment guidelines. Clin Infect Dis 2022; 74(Suppl_2):S152. doi:10.1093/cid/ciac030
- C Alessio and P Nyirjesy. Management of resistant trichomoniasis. Curr Infect Dis Rep 2019; 21:31. doi:10.1007/s11908-019-0687-4
- AP Ghosh et al. In vitro study of the susceptibility of clinical isolates of Trichomonas vaginalis to metronidazole and secnidazole. Antimicrob Agents Chemother 2018; 62:e02329. doi:10.1128/aac.02329-17
- LA Vodstrcil et al. Male-partner treatment to prevent recurrence of bacterial vaginosis. N Engl J Med 2025; 392:947. doi:10.1056/nejmoa2405404
- ACOG. Concurrent sexual partner therapy to prevent bacterial vaginosis recurrence. Obstet Gynecol 2025; 146:e111. doi:10.1097/aog.0000000000006102
- H Yusuf and M Trent. Management of pelvic inflammatory disease in clinical practice. Ther Clin Risk Manag 2023; 19:183. doi:10.2147/tcrm.s350750
- FJ Chevalier et al. Syphilis: a review. JAMA 2025; 334:1927. doi:10.1001/jama.2025.17362
- EW Hook III et al. One dose versus three doses of benzathine penicillin G in early syphilis. N Engl J Med 2025; 393:869. doi:10.1056/nejmoa2401802
- T Klementová et al. Cefixime versus benzathine penicillin G for the treatment of early syphilis-a randomized, controlled open label trial. J Antimicrob Chemother 2025; 80:2654. doi:10.1093/jac/dkaf268
- CDC. Availability of STI testing and treatment products. March 10, 2026. Available at: https://bit.ly/4cizH2j. Accessed April 9, 2026.
- In brief: Alternatives to Bicillin L-A. Med Lett Drugs Ther 2026; 68:7.
- ASHP. Current drug shortages: penicillin G benzathine. March 12, 2026. Available at: https://bit.ly/48lwQV0. Accessed April 9, 2026.
- M Collins-Ogle and BC Herold. Developments in genital herpes: progress in prevention and treatment. Curr Opin Infect Dis 2026; 39:61. doi:10.1097/qco.0000000000001177
- A Wald et al. Comparative efficacy of famciclovir and valacyclovir for suppression of recurrent genital herpes and viral shedding. Sex Transm Dis 2006; 33:529. doi:10.1097/01.olq.0000204723.15765.91
- L Corey et al. Once-daily valacyclovir to reduce the risk of transmission of genital herpes. N Engl J Med 2004; 350:11. doi:10.1056/nejmoa035144
- Y Panahi et al. The efficacy of topical and oral ivermectin in the treatment of human scabies. Ann Parasitol 2015; 61:11.
- RL Woosley et al. QTdrugs list, AZCERT, Tucson, AZ. Available at: www.crediblemeds.org. Accessed April 9, 2026.
- Inhibitors and inducers of CYP enzymes, P-glycoprotein, and other transporters. Med Lett Drugs Ther 2023 January 25 (epub). Available at: www.medicalletter.org/downloads/CYP_PGP_Tables.pdf.
The Medical Letter, Inc. does not warrant that all the material in this publication is accurate and complete in every respect. The Medical Letter, Inc. and its editors shall not be held responsible for any damage resulting from any error, inaccuracy, or omission.